First-in-class Pharmacological Treatment of AMI
Epoqe Pharma is a pre-clinical-stage biopharmaceutical company focused on the development of novel treatments for serious cardiovascular diseases.
Acute Myocardial Infarct
Acute obstruction of the coronary blood flow limits oxygen supply to the heart muscle and induce myocardial infarction (also known as heart attacks). This promotes cardiac ischemia, oedema, dysfunction and increases the risk of ischemic heart disease and heart failure. Ischemic heart disease feeds into a cardiometabolic disease acceleration loop. Ischemic heart disease is #1 global cause of death, disability and human suffering with costs of 108 bn$ annually (Global Burden of Disease, 2022).
- Fast therapeutic intervention to reperfuse tissue is essential for survival and positive prognosis
- No pharmacological option for the rescue of heart function currently exists
- There are more than 3 mio cases of AMI annually (1 mio in US only) and the estimated out of hospital mortality is 30-50%
- Currently, patients reaching the hospital for acute surgery foresee 10% mortality risk, 10-40% risk of secondary heart failure and 5-10% risk of 30-days readmission.
Mission & vision statements
Vision
Our vision is to prevent death and disability for millions of people worldwide suffering from serious cardiovascular diseases and to reduce the burden for individuals, families and society.
Mission
Our mission is to develop PPs for the treatment of acute myocardial infarct (AMI) and additional acute and chronic cardiovascular diseases.
The Epoqe Pharma Concept
Our concept is utilizing a natural heart rescue mechanism, a strong scientific rationale, solid IP, and a novel mode of action. The Epoqe pharma PPs are analogs of a native peptide. PPs induce previously unseen heart function recovery (45-86%), and reduces the infarct size (50-66%) in pre-clinical proof-of-concept studies in minipig (ischemia and reperfusion) and rat (ischemia without reperfusion).
PPs and the native peptide induce biological actions that are highly conserved among species. The scientific rationale is based on well established biology, a well characterized receptor population and preferred PP actions in ischemic heart tissue.
We believe PPs induce both a biological reperfusion and a direct protection of ischemia- and inflammation-induced cardiomyocyte injury.
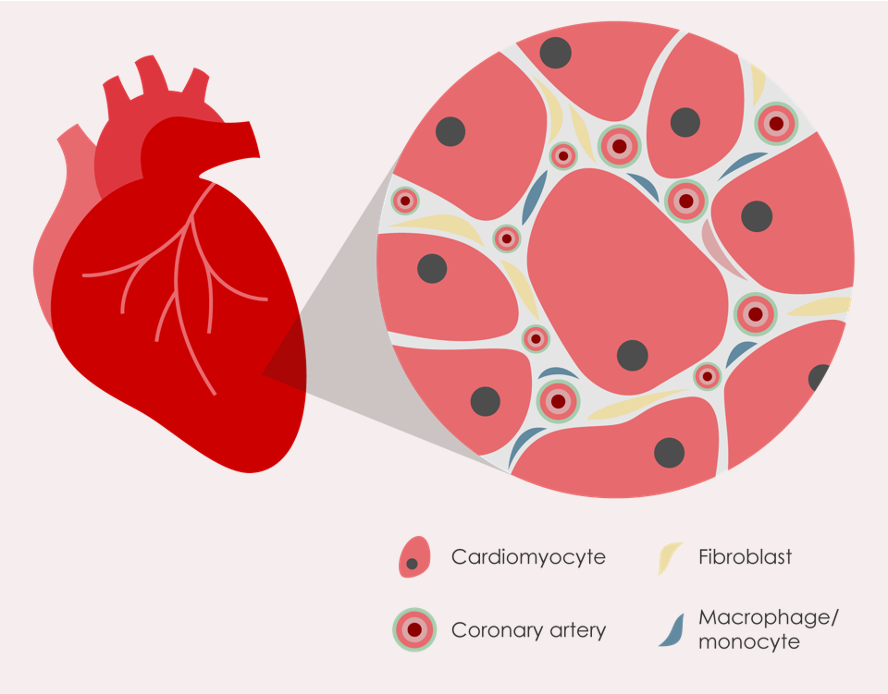
CIRCUMVENTION
PPs increase blood supply to the infarcted area presumably through direct vasodilation and vascular reserve recruitment
REGENERATION
PPs likely induce local angiogenesis during cardiac ischemia which facilitates a sustained 'biological reperfusion' effect
PRESERVATION
PPs likely induce direct protection of cardiomyocytes from ischemia- and inflammation-induced injury
News
November 2023: Press release
July 2023: Press release
Team

ANETTE SAMS, PHD
CEO, CSO & Co-founder

JENS BUKRINSKI, PHD
CTO & Co-founder
Supported by



